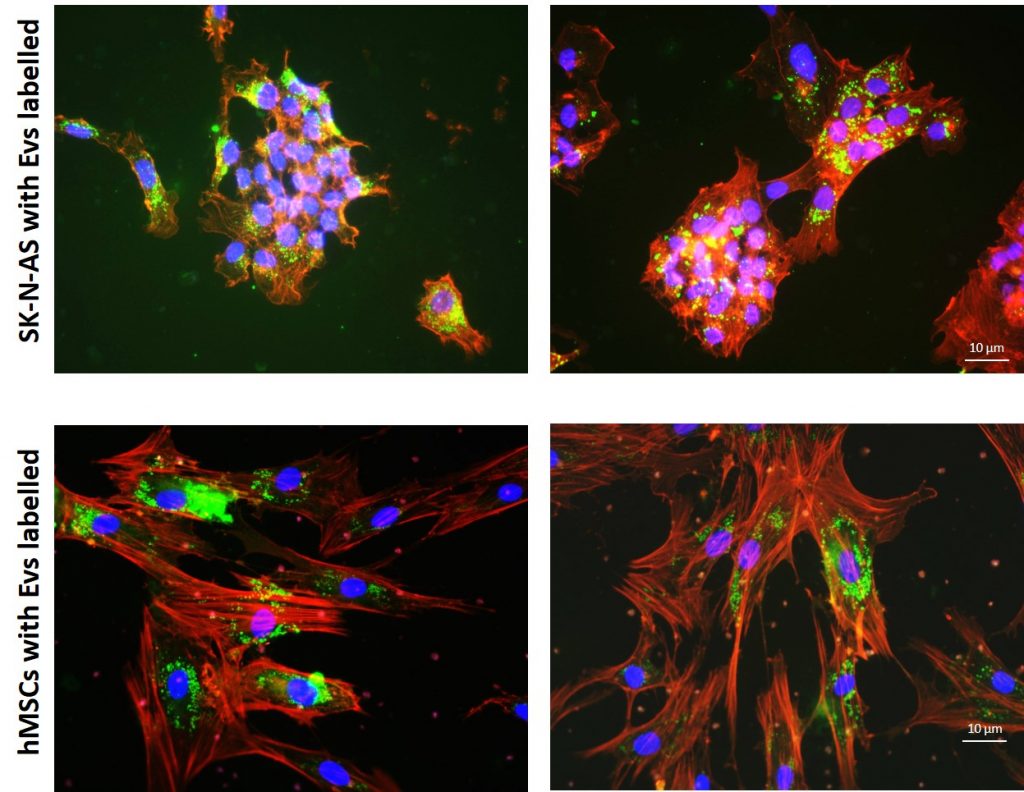
Tumor hypoxia stimulates release of extracellular vesicles (EVs) that facilitate short- and long-range intercellular communication and metastatization. Albeit hypoxia and EVs release are known features of Neuroblastoma (NB), a metastasis-prone childhood malignancy of the sympathetic nervous system, whether hypoxic EVs can facilitate NB dissemination is unclear.EVs derived from hypoxic NB cells were more potent than their normoxic counterpart in inducing NB cells migration and colony formation. We also focused on the effects of EVs in metastatic niches formation in vivo. Intravascular injection within the zebrafish embryo allows a non-invasive visualization of EVs dispersion, uptake, and interactions with host cells. We have demonstrated that EVs released under hypoxic conditions promote angiogenesis and are more easily internalized by endothelial cells than those purified from normoxic cells. Furthermore, we have proved via microscopy imaging and cell sorting that the group injected with hypoxic EVs had higher numbers of macrophages. We showed that hypoxic EVs induce i. angiogenesis in vivo, required for tumor and metastasis growth, and ii. increased number of macrophages, a major contributor to the tumor microenvironment which produce growth factors and chemokines promoting tumor angiogenesis. Our data demonstrate that hypoxic EVs can modify the behavior of recipient cells in the CHT, as a potential metastatic site, to prepare a ‘fertile soil’ for cancer cells colonization.